Sugars
Simple Sugars
Sugar is a type of carbohydrate that takes on many forms in nature. Yeasts can only digest what are called simple sugars which are the most basic type of sugar molecule, common examples include glucose, fructose, and sucrose, the latter we know as table sugar. Each of their chemical structures is across the page, which are relatively simple and short.
Simple sugars exist in virtually all fruits as well as some plants - notably sugar cane. Thus, most fruit will easily ferment on its own. If you lightly crush a few grapes and leave them in a loosely covered container, a few days later it will have turned to wine (this seemingly magical phenomenon is probably one reason why wine is affiliated with divinity).
You can read more read about sugar, how it is made and the different types here.
Lactic Acid Fermentation - This is another type of fermentation that it has nothing to do with making booze. It occurs when bacteria in dairy, fruits, and vegetables, or muscles cells in animals, find themselves in an environment with no available oxygen. As a last resort, these organisms ferment any available simple sugars to use as an alternative energy source and create lactic acid in the process. This method is responsible for producing many of your favorite fermented food products like yogurt, pickles, and kimchi. It is also why your muscles are sore after exerting yourself while exercising.
How it Works
Fermentation is the one and only way booze can be created, and that is why we love it. It occurs between yeast and sugar. Under the right conditions - moist and warm is ideal - the yeast will consume the sugar, converting it into energy and excreting out alcohol (specifically, ethanol) in the process. Yeast will keep on devouring sugar, and thus producing alcohol, until all the available sugars are consumed, or the alcohol levels reach around 15-17% ABV, at which point the yeast can no longer survive and die off. To get the proof any higher, distillation is needed.
In addition to alcohol, yeast also expels carbon dioxide, which is what bakers utilize these to make bread rise and some brewers still rely on for carbonation in their beer.
A third byproduct of fermentation is the creation of congeners, which are a catch-all term for a variety of organic compounds that supply alcoholic beverages with flavor. Because ethanol on its own doesn’t taste like much. Brewers, winemakers, and distillers rely on congeners and employ a variety of techniques to manipulate the kinds that are created while maximizing the alcoholic output.
On this page, we'll look at the inner workings of fermentation . Biology and chemistry awaits!
Fermentation
Yeast + Sugar = Alcohol

Yeast
Yeast is type of single-celled fungus, not a bacteria, and a relative of mold. It exists pretty much everywhere - there are yeasts floating around aimlessly in the air around you - and there are hundreds of different species. Some cause infection, some spoil food, and some make alcohol. Most alcohol-making yeasts are members of the Saccharomyces genus. The species S. cerevisiae specifically supplies the strains commonly known as brewer’s and baker’s yeast.
All types, or strains, of yeast behave differently in different environments. Which strain a booze producer uses depends on the results they are after. Some producers use natural or "wild" yeasts, meaning they allow whatever yeasts are hanging around nearby to do the job, though most carefully select yeasts, or cultivate their own strains for optimal control of the flavor profile they are after. For those producers, yeast is a closely guarded proprietary commodity.


Glucose

Fructose

Sucrose
(combination of glucose and fructose)
These are three examples of simple sugars, consisting of one or two molecules, which yeast can easily digest.

Aspergillus oryzae (koji) growing on rice
Courtesy of Wikimedia Commons
A Bit More About Esters
Esters play an integral role in the flavor profile of many spirits so I thought I'd spend just a bit more time on them.
As I said above, esters are a class of organic compound that have varying fruity and floral aromas. Esters can be created during fermentation when an alcohol molecule and an acid molecule combine - both of which are plentiful of in a fermentation batch.
What I think is really cool about this is that two compounds with unappealing or even repulsive qualities can yield one that's delicious. My favorite example is with the ester called Ethyl Butyrate, which I found described on cocktailwonk.com, a wonderful site for cocktail/spirits nerds, written by Matt Pietrek. In his words: “Ethyl Butyrate is formed when ethyl alcohol molecules combine with butyric acid. Butyric acid by itself has the smell of human vomit. But combine it with ethyl alcohol, and the resulting Ethyl Butyrate molecule smells of fruit and pineapple."
So, human vomit flavor: eliminated. Pineapple flavor: created. Esters FTW!
Congeners
As mentioned above, congener is an umbrella term for a collection of organic chemical compounds created during fermentation that supply spirits, wine, and beer with their particular flavors. Congeners make up for about 25% of a fermentation batch, the rest of which is alcohol (ethanol) and water. Some taste good, some taste bad, and some are downright toxic. Here are some examples of common congeners:
-
Methanol - A toxic type of alcohol that I like to call "bizarro ethanol". Too much of it will make you go blind. Though it is ok in small amounts and is present in most spirits.
-
Acetaldehyde - A type of aldehyde (which is an oxidized alcohol) that has a sharp, metallic-y green apple flavor.
-
Acetone - A ketone that smells like nail polish remover.
-
Esters - A class of organic compounds that have varying fruity and floral aromas. You might say they are the brass ring of congeners. More on these below.
-
Sulfurous Compounds - A collection of compounds containing sulfur that have off-putting aromas of eggs and cabbage. Copper helps to strip these compounds out, which is why it is the go-to material for constructing stills.
-
Fusel Oils - A group of heavy, oily and generally unpleasant alcohols: butanol, propanol and amyl alcohol. Fusel is a German word for bad booze or rotgut. However, in small amounts, these can benefit a spirit's body.
-
Acetic Acid - An acid that is best known for giving vinegar its familiar penetrating aroma.
Congeners can be removed or retained during distillation, and removed and/or modified post-distillation. Further details are on the Distillation and Maturation Pages.
Esters Haiku
By Seth Wright, Brewer at Wachusett Brewing Co. in Westminster, Ma.
I found this poking around online and couldn't resist adding it here. It's about brewing beer, but the sentiment still applies:
Yeast stress shall dictate
Levels of isoamyl acetate
Brewers salivate
Human Saliva
The third way to convert starch into fermentable sugars is the weirdest, but also the simplest. Chewing it! Human saliva contains the enzymes that do the trick. In fact, there’s a traditional drink in South America called Chicha which is made by chewing corn. Strange to be sure, but if I had no other option, I'd definitely I'd give it a shot.
It probably goes without saying that this method, thankfully, is not used for mass production.
Breaking Down Starches into Simple Sugars
Starches can be broken down into simple sugars by enzymes called amylase or diastase. Accessing these enzymes is done in one of three ways:
Malting
As luck would have it, many grains already contain the amylase necessary to break down their starches, it just has to be activated. This is done by a method called malting, steeping the grain in water which convinces it that it’s time to germinate. This effectively releases the enzymes and the breakdown begins. In nature, the purpose of these enzymes is to create food and energy for the growing plant. But before actual germination can take place, the grains are dried, encapsulating the sugars for fermentation.
The most commonly malted grain is barely because it contains the highest concentration of amylase. Barley that has been malted is called, as you might imagine, malted barley, or simply "malt".
Malting is the traditional approach in the west for starch breakdown and is employed in the majority of whiskey and beer production.
Koji
Koji, aka Aspergillus oryzae, is a particular species of fungus that grows as a mold which contains the precious amylase. In this method, the koji is added to whatever is being fermented. This inoculates the batch with amylase and facilitates the breakdown.
This method was developed in the far east and is used to make sake, shochu, and other traditional Asian alcoholic beverages, though it can be used anywhere. Some Canadian whiskeys use koji, for example.
Koji fungus is usually cultivated with moldy rice cakes called "qus". These often have yeast added to them so when introduced to a batch, the breakdown of starch and fermentation can happen simultaneously.
Congener Creation and Manipulation
The kind of congeners created during fermentation will largely depend in part on the strain of yeast being used. Though producers have other methods at their disposal which influence the result as well. Here are a few of the biggies:
Time
A shorter fermentation - 3-4 days - will generally create fewer congeners and, thus, a milder tasting product because there will be less time for congeners to develop and react with one another. On the flip side, longer fermentations - 5 days and beyond - will develop more intense and diverse flavors. In extreme cases, fermentation will go on for 1-3 weeks, or even longer. In these cases, bacteria will eventually begin to form which adds another congener producing variable to the mix that further expands the flavor range - for better or worse.
Temperature and Oxygen
Warmer temperatures will increase the rate of fermentation and thus the development of congeners, while lower temperatures stretch out the process offering more control of microbial growth and avoiding the potential for spoilage. Access to oxygen, or lack thereof, is also key in how the yeasts behave. The result depends on the strain of yeast and the style of spirit being made.
Fluctuating the temperature and oxygen levels will cause the yeasts behave in abnormal ways, producing different congeners, notably esters. This is commonly known as intentionally “stressing” the yeast. If done wrong it can ruin a batch, but if done right it can give spirits wonderful depth and complexity. Note, for beer-makers stressed yeast is typically a bad thing because all the congeners created will remain in the final product. Brewers call these off flavors. But remember, distillers have the ability to remove, purify, and/or modify, congeners during and post-distillation.
Additives: Backset, Bacteria, Dunder
Sometimes other ingredients will be manually added to a fermentation batch to cultivate congeners or maintain a certain environment. One example of this is famously done in American whiskey production where it is known as the sour mash process. Dead yeast cells leftover in the still from a previous batch, also known as the backset, are introduced to the next fermentation batch to creates an acidic environment which is beneficial to the yeast. This method is also traditional with many Jamaican rums, where the backset is called "dunder ". Dunder is sometimes stored in pits called dunder pits or "muck" pits which helps to cultivate acid-producing bacteria that can be turned into esters when the dunder is added to the fermentation batch. This latter technique yields some of the most unique flavors you'll ever encounter in a spirit.
Starches - Complex Sugars
Another form that sugar takes is as a starch, such as the ones found in grains like corn, rye or wheat. Of course, grains like these are relied upon are the bases of two of life's most indispensable delights: beer and whiskey.
But grains will not ferment naturally on their own, because starches are too complex for the yeast to digest. They are made up of multiple simple sugars strung together, this is illustrated across the page by two the most common starch molecules: amylose and amylopectin. As you can see, the chains are much longer and more elaborate. To ferment grains, starch needs to be separated into smaller easily-consumable simple sugars first.
Luckily, there are ways to do this, which we'll look at below.

Amylose
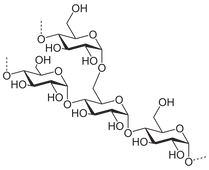
Amylopectin
These are starch chains made up of several simple sugar molecules (glucose, specifically) strung together. Yeast cannot digest these naturally, they need to be broken apart first.
Some Common Esters Found in Booze:
Propyl acetate (Pears)
Octyl acetate (Oranges)
Isoamyl acetate (Banana)
Butyl acetate (Apple)
Methyl trans-cinnamate (Strawberry)
Ethyl cinnamate (Cinnamon)

